|
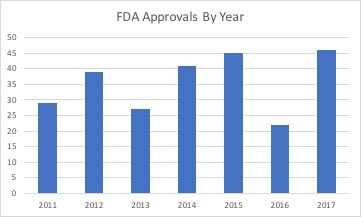
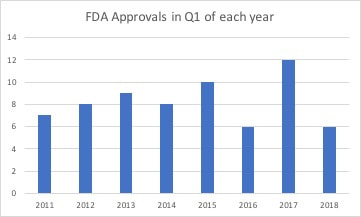
Much was written about the increase in drug approvals by the FDA in 2017. Some speculated it showed a relaxation of standards to bring drugs to market at a quicker pace. But if you look at the trend in the number of drugs approved by the FDA last year, it was similar to 2015 and just a little higher than 2012 and 2014. So we likely just saw an increase that was within the normal behavior of the FDA. The increase in approvals was likely a byproduct of the FDA clearing a backlog of orphan drug requests as the agency began to implement FDA Commissioner Scott Gottlieb's goal of improving the process for reviewing drugs. So if there was a change in philosophy at the FDA on drugs reviews, then we would expect to see an increase in 2018. Only one quarter has passed, so it is really too early to determine what the final volume of approvals might be. But if we look at the number of approvals for the first quarter of each year, back to 2011, it suggests this may be a year with a depressed number of approvals. The beginning of 2013 suggested a robust year for approvals, but ended as the second lowest total. In 2012 and 2014 the year started off modestly, but ended robustly. So the pace of approvals could pick up in the last three quarters to the year, but the current volume suggests a total similar or slightly higher than 2016. You can stay up-to-date with drugs in development with the Prescribe Right Pharmaceutical Pipeline Tracker. Comments are closed.
|
Stay informed, subscribe to the
Prescribe Right Pharmaceutical Pipeline Tracker Latest Tweets from Prescribe Right
Archives
July 2023
|
Services |
Company |
Support |
© COPYRIGHT 2015. ALL RIGHTS RESERVED.
|


 RSS Feed
RSS Feed